Researchers have been able to combine fluorescence observation methods with a microscope equipped with a high numerical aperture objective. With the emergence of biological specific fluorescent protein (such as green fluorescent protein, GFP), fluorescent observation also began to appear in stereo microscope.
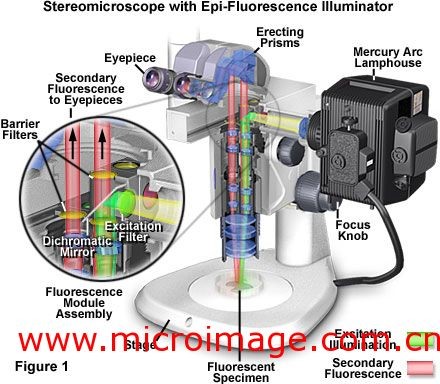
Due to the large field of vision and working distance of stereoscope, researchers can distinguish the organisms expressing fluorescent protein, such as nematodes, zebrafish, oocytes, mature insects and so on. At the same time, researchers can use tweezers, droppers or other micromanipulation devices to process samples. In addition, under the stereoscope, the stereoscopic fluorescent lighting is more conducive to the observation and recording of samples.
The fluorescent illuminator of the stereoscope is the same as that of the composite microscope. In the fluorescent illuminator, xenon lamp or mercury lamp is used as the light source, and a lamp chamber is covered, and the lamp chamber is connected with the microscope body through a vertical illuminator. As shown in Figures 1 and 2, the vertical illuminator is located between the zoom body of the fuselage and the observation tube. Next, we take the Nikon smz1500 stereoscope as an example to introduce the light path of lower stereoscopic fluorescence. The light emitted by the light source passes through the converging lens to the excitation light filter in the color filter block group, where only a part of the light in the light source (a certain wavelength) can pass through the filter. The light passing through the filter is reflected by the semi transparent and semi reflective mirror, passes through the zoom lens group and objective lens at the lower part of the fuselage, and reaches the sample. A semi transparent and semi reflective mirror has the property of reflecting light smaller than a specific wavelength and transmitting light larger than a specific wavelength.
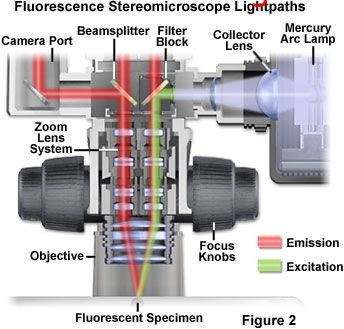
The light passing through the objective lens has been inverted cone to irradiate the sample and excite the corresponding fluorescent substance. The emitted fluorescence passes through the objective lens, the intermediate aneuploid and reaches the emission light filter. Here, a part of the light will be filtered out, and only the excited fluorescence will pass through the emission light filter. Figure 1,2 shows the optical path diagram. In the light path on the left side, the emitted fluorescence directly passes through the emitting light filter and then enters the camera or eyepiece observation tube; in the light path on the right side, the emitted light first passes through the semi transparent and semi reflective mirror, and then passes through the emitting light filter and enters the eyepiece observation tube.
Figure 2 and 3 are the schematic diagram of fluorescent filter block group. Each filter block group has a semi transparent and semi reflective mirror, an excitation light filter and two emission light filters. As described earlier, the light emitted by the light source passes through the excitation light filter and enters the filter block group, which is reflected by the semi transparent and semi reflective mirror; the emitted fluorescence passes through the emission light filter and enters the eyepiece or camera. In this combination, the semi transparent and semi reflective mirror and the emission light filter on the left side are fixed on the shelf, while the emission light filter on the right side is installed in a removable filter frame. As long as the screws are loosened, the right filter frame can be removed, and then the semi transparent and semi reflective mirror can be replaced as needed. When replacing, pay attention not to apply the fixed glue to the semi transparent and semi reflective mirror, and wear gloves to avoid leaving fingerprints on the mirror.
The stereo microscope can be equipped with three fluorescent filter blocks and one filter free block for bright field observation. During observation, the position of the color filter block can be adjusted by pulling the lever, and each color filter block is equipped with a different logo for easy selection.

A variety of filter combinations available for Nikon in table1. These filters cover most of the wavelength range of fluorescence excitation / emission (the excitation band is 380nm-510nm), and are suitable for biological and industrial research. At the same time, the following combinations are also suitable for the study of green fluorescent protein variants, such as cyan and blue fluorescent protein. In the observation of living cells, choosing the appropriate combination of color filters will improve the signal intensity. In addition, considering the autofluorescence of chlorophyll, the filter set specially designed for plant observation should be selected.
Combination of fluorescent filter for body mirror:
Excitation group |
Excitation filter |
Dichroic mirror |
Emission filter |
Blue GFP/DAPI |
379-401 nm |
420 nm (DCLP) |
435-485 nm |
Cyan (EGFP) GFP |
426-446 nm |
455 (DCLP) |
460-500 nm |
Endow GFP band-pass |
HQ 450-490 nm |
Q 495 (LP) |
500-550 nm |
Endow GFP long-pass |
HQ 450-490 nm |
Q 495 (LP) |
500 nm (LP) |
TRITC (DsRed) |
HQ 530-560 nm |
Q 570 (LP) |
590-650 nm |
Yellow GFP band-pass |
HQ 490-510 nm |
Q 515 (LP) |
520-550 nm |
In addition, the environmental humidity will also affect the bandpass of the filter. In order to prolong the service life of the filter, the filter should be stored in the dryer or placed in a sealed box with a dryer. At the same time, we should reduce the exposure time of the filter. If we don't observe, we should turn off the light gate of the light source to protect the filter. When cleaning the filter, use ear ball, camel hair brush, or oil-free gas pressurizer to clean through dry air. Never use mirror cleaning paper. They will scratch the coating on the filter.
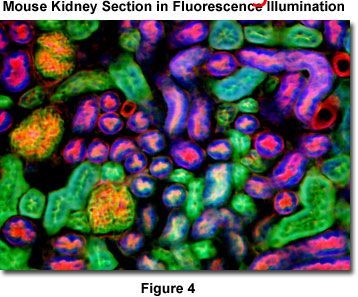
Taking Nikon smz1500 as an example, the magnification of this stereoscope is 4x to 540x, which is close to that of a composite microscope. With such a stereoscope, researchers can observe a large volume of live samples or thin sections labeled with fluorescence. Figure 4 is a three color fluorescent staining of mouse kidney sections. The dyes used were DAPI, Alexa fluor 488 WGA, and Alexa fluor 568. The corresponding filters were blue GFP / DAPI, end GFP bandpass, and TRITC DsRed in Table 1. This image shows that the stereoscope has the ability to observe the sample of compound microscope.
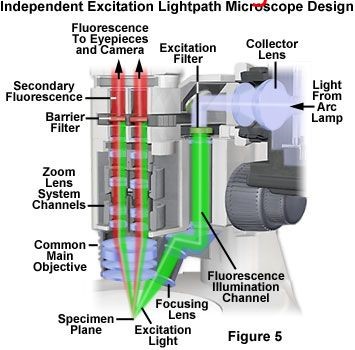
Other microscope manufacturers have different fluorescent lighting designs, such as Figure 5, which is one of the more mainstream models. In this design, there is a fluorescent illumination channel independent of the imaging light path. The light from the light source first passes through the excitation light filter, and then passes through the channel behind the host to the lens group at the bottom, which transmits the light to the sample. This design ensures that no matter what magnification, the excitation light will directly irradiate the sample, and the range, intensity and black background will not change with the magnification. After the emitted fluorescence reaches the aneuploid through the objective lens, it passes through the emission light filter and enters the eyepiece for observation or camera. The main advantage of this design is that it eliminates the semi transparent and semi reflective mirror, and the combination of filters is variable, so researchers can choose a wide range. However, it also brings some problems, for example, the inexperienced operator may not observe the fluorescence due to selecting the wrong filter combination.
The intense ultraviolet light produced by mercury lamp can damage the retina of the observer. In order to avoid this accident, many manufacturers use protection facilities to filter out the ultraviolet radiation on the sample and stage. At the same time, there are many other protection measures, such as adding filter in the light path. A shutter is usually installed between the light box and the vertical illuminator in the stereoscopic fluorescent mirror. When the sample is not observed or photographed, the shutter will be closed to protect the sample from long-term ultraviolet radiation. Sometimes in the case of high magnification flat field achromatic objective (1.6x to 2.0x) with low zoom ratio, reflection and hot spots may appear in the lower part of the field of vision. This illusion usually disappears when the zoom ratio is raised. Moreover, when using low magnification objective (0.5 x or 1.0 x) and high magnification low correction objective (achromatic objective, flat field achromatic objective), this false image will not appear. Using stereoscope, you can observe many kinds of samples, such as table2, from biology to industrial manufacturing.
Application of fluorescence in body mirror
Area |
Applications |
Biology |
Gene expression, cell classification, anatomy, developmental process, eye and muscle research |
Botany |
Plant cells, tissue autofluorescence, encourage samples, parasites |
Pharmacology |
Capillary flow, drug, observation |
Hydrology |
Water quality, cell structure, membrane analysis |
Agricultural Economics |
Seed observation, gene expression, transgenic |
Electronics |
Solder paste, epoxy resin analysis, display coating, integrated circuit polymer casting |
Semiconductor |
Photoresist pollution, foreign particles, processing error |
Polymer |
Foreign particles, voids, bead analysis, nonpolymerization zone |
Metal manufacturing |
Cracks and surface defects, contamination, welding control, fracture analysis |
Material Science |
Crack, welding, carbon bond, fracture, location research |
| Forensic medicine | Textile fibers, body fluids, fingerprints, bank notes, fakes |
Papermaking |
Fiber coating and inclusion |